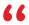- Contact 0870 350 7767
- |
- Advertise
New Certification for our Managing Director, Jon Jones
 News and PR from Cranage Veritas Limited - Published 28 July 2020
We are delighted to announce that our Managing Director, Jon Jones, has been approved as a Factory Follow-Up Inspector for Eurofins E&E North America (Eurofins MET Labs).
News and PR from Cranage Veritas Limited - Published 28 July 2020
We are delighted to announce that our Managing Director, Jon Jones, has been approved as a Factory Follow-Up Inspector for Eurofins E&E North America (Eurofins MET Labs). "I'm proud to have been approved as a Factory Inspector with Eurofins MET Labs, and am looking forward to strengthening our working relationship with them. It's a significant development for us at Cranage Veritas as we enhance our local NRTL business; this is an essential service for customers looking to gain NRTL Certification, and I'm delighted I can now evaluate factory processes as part of this. I'm eager to build relationships with factories in Ireland and develop strong working connections."
To find out more, or for a no obligation discussion about a factory inspection at your site, contact Jon Jones: [email protected].
+353 061 633 630
Other announcements from Cranage Veritas Limited
-
Cranage Veritas - Medical Device Services
View our market access services for medical devices, including certification for North American and Canadian Markets, FDA Approval, CE Mark Assistance and CB Certification.
07 Mar 2022
-
Our New Website is Now Live!
Cranage Veritas is pleased to announce that our website has been refreshed! Now featuring our full range of services, as well as useful information on product marking and compliance engineering.
22 Feb 2022
-
Cranage Veritas Christmas Newsletter
We would like to send our customers our best wishes for the festive season and we look forward to working with you in 2022.
16 Dec 2021
-
The Medical Device Consultancy service from Cranage Veritas
Are you looking for a simple and effective route to market for your medical device?
03 Dec 2021
-
Supporting manufacturers and importers with the UKCA mark transition
At Cranage Veritas we can assist you with the transition to the UKCA mark that is proposed for implementation in the UK on the 1st January 2021.
06 Oct 2020
-
Do you need help to complete a product risk assessment?
Our handy help sheet has more information about the importance of a product risk assessment and how our team of expert engineers can assist you.
20 Aug 2020
-
Technical File Review
Do you need to review your CE Mark? Do you need to know more about the importance of a Technical File or Technical File review? Our handy information sheet can help.
04 Aug 2020
-
Cranage Veritas Announcement
Cranage veritas announce their managing Director has been approved as a factory Follow-up inspector by Eurofins E&E north America (Eurofins Met Labs)
28 Jul 2020
-
CRANAGE VERITAS CELEBRATES THE SUCCESS OF ITS MANAGING DIRECTOR
LIMERICK BASED BUSINESS, CRANAGE VERITAS, CELEBRATES THE SUCCESS OF ITS MANAGING DIRECTOR WHO HAS BEEN APPOINTED AS THE ONLY LOCAL TECHNICAL REPRESENTATIVE (LTR) IN IRELAND.
16 Jun 2020